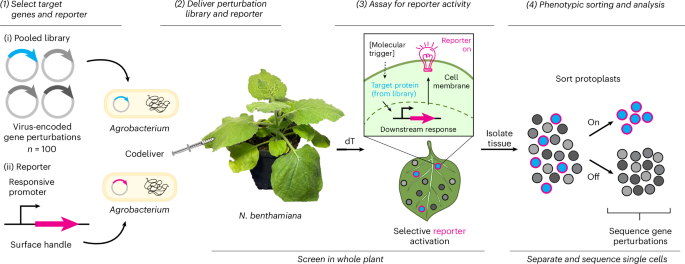
A single-cell screening platform accelerates functional genetics in plants – Nature Biotechnology
Gómez-Gómez, L. & Boller, T. FLS2: an LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol. Cell 5, 1003–1011 (2000).
Google Scholar
Bock, C. et al. High-content CRISPR screening. Nat. Rev. Methods Primers 2, 8 (2022).
Google Scholar
Chong, Z.-S., Wright, G. J. & Sharma, S. Investigating cellular recognition using CRISPR/Cas9 genetic screening. Trends Cell Biol. 30, 619–627 (2020).
Google Scholar
Ngou, B. P. M., Ding, P. & Jones, J. D. G. Thirty years of resistance: zig-zag through the plant immune system. Plant Cell 34, 1447–1478 (2022).
Google Scholar
Jores, T. et al. Identification of plant enhancers and their constituent elements by STARR-Seq in tobacco leaves. Plant Cell 32, 2120–2131 (2020).
Google Scholar
Gorjifard, S. et al. Arabidopsis and maize terminator strength is determined by GC content, polyadenylation motifs and cleavage probability. Nat. Commun. 15, 5868 (2024).
Google Scholar
Arndell, T. et al. Pooled effector library screening in protoplasts rapidly identifies novel Avr genes. Nat. Plants 10, 572–580 (2024).
Google Scholar
Lau, W. & Sattely, E. S. Six enzymes from mayapple that complete the biosynthetic pathway to the etoposide aglycone. Science 349, 1224–1228 (2015).
Google Scholar
McClune, C. J. et al. Discovery of FoTO1 and taxol genes enables biosynthesis of baccatin III. Nature 643, 582–592 (2025).
Google Scholar
Zürcher, E. et al. A robust and sensitive synthetic sensor to monitor the transcriptional output of the cytokinin signaling network in planta. Plant Physiol. 161, 1066–1075 (2013).
Google Scholar
Hertig, C. W. et al. Genome-wide characterization of two-component system elements in barley enables the identification of grain-specific phosphorelay genes. BMC Plant Biol. 25, 209 (2025).
Google Scholar
Alamos, S. et al. Multiplexed profiling of transcriptional regulators in plant cells. Nat. Biotechnol. https://doi.org/10.1038/s41587-025-02880-w (2025).
Carlson, E. D., Rajniak, J. & Sattely, E. S. Multiplicity of the Agrobacterium infection of Nicotiana benthamiana for transient DNA delivery. ACS Synth. Biol. 12, 2329–2338 (2023).
Google Scholar
Sainsbury, F., Thuenemann, E. C. & Lomonossoff, G. P. pEAQ: versatile expression vectors for easy and quick transient expression of heterologous proteins in plants. Plant Biotechnol. J. 7, 682–693 (2009).
Google Scholar
Cumby, N., Edwards, A. M., Davidson, A. R. & Maxwell, K. L. The bacteriophage HK97 Gp15 moron element encodes a novel superinfection exclusion protein. J. Bacteriol. 194, 5012–5019 (2012).
Google Scholar
Hull, R. & Plaskitt, A. Electron microscopy on the behavior of two strains of alfalfa mosaic virus in mixed infections. Virology 42, 773–776 (1970).
Google Scholar
Adams, R. H. & Brown, D. T. BHK cells expressing Sindbis virus-induced homologous interference allow the translation of nonstructural genes of superinfecting virus. J. Virol. 54, 351–357 (1985).
Google Scholar
Cassells, A. C. & Herrick, C. C. Cross protection between mild and severe strains of tobacco mosaic virus in doubly inoculated tomato plants. Virology 78, 253–260 (1977).
Google Scholar
Julve, J. M. et al. A coat-independent superinfection exclusion rapidly imposed in Nicotiana benthamiana cells by tobacco mosaic virus is not prevented by depletion of the movement protein. Plant Mol. Biol. 81, 553–564 (2013).
Google Scholar
Lindbo, J. A. TRBO: a high-efficiency tobacco mosaic virus RNA-based overexpression vector. Plant Physiol. 145, 1232–1240 (2007).
Google Scholar
Rabindran, S. & Dawson, W. O. Assessment of recombinants that arise from the use of a TMV-based transient expression vector. Virology 284, 182–189 (2001).
Google Scholar
Avesani, L. et al. Stability of potato virus X expression vectors is related to insert size: implications for replication models and risk assessment. Transgenic Res. 16, 587–597 (2007).
Google Scholar
Birnbaum, K. et al. Cell type-specific expression profiling in plants via cell sorting of protoplasts from fluorescent reporter lines. Nat. Methods 2, 615–619 (2005).
Google Scholar
Harkins, K. R. & Galbraith, D. W. Factors governing the flow cytometric analysis and sorting of large biological particles. Cytometry 8, 60–70 (1987).
Google Scholar
Los, G. V. et al. HaloTag: a novel protein labeling technology for cell imaging and protein analysis. ACS Chem. Biol. 3, 373–382 (2008).
Google Scholar
Li, C. et al. Glycosylphosphatidylinositol-anchored proteins as chaperones and co-receptors for FERONIA receptor kinase signaling in Arabidopsis. eLife 4, e06587 (2015).
Google Scholar
Vlahos, A. E. et a.l A complete suite for compact programming of protein secretion. Preprint at bioRxiv https://doi.org/10.1101/2023.10.04.560774 (2025).
Xu, M., Du, Q., Tian, C., Wang, Y. & Jiao, Y. Stochastic gene expression drives mesophyll protoplast regeneration. Sci. Adv. 7, eabg8466 (2021).
Google Scholar
Jeyaprakasam, J. & Tennyson, J. Isolation and transfection of Nicotiana benthamiana mesophyll protoplasts. Methods Mol. Biol. 2973, 251–260 (2025).
Google Scholar
Hwang, I., Sheen, J. & Müller, B. Cytokinin signaling networks. Annu. Rev. Plant Biol. 63, 353–380 (2012).
Google Scholar
Rashotte, A. M. et al. A subset of Arabidopsis AP2 transcription factors mediates cytokinin responses in concert with a two-component pathway. Proc. Natl. Acad. Sci. 103, 11081–11085 (2006).
Google Scholar
Zhang, J. et al. AtSOFL1 and AtSOFL2 act redundantly as positive modulators of the endogenous content of specific cytokinins in Arabidopsis. PLoS ONE 4, e8236 (2009).
Google Scholar
Voges, M. Molecular Principles to Engineer Plant Microbiomes. PhD thesis, Stanford University (2019).
Han, Z.-F., Hunter, D. M., Sibbald, S., Zhang, J.-S. & Tian, L. Biological activity of the tzs gene of nopaline Agrobacterium tumefaciens GV3101 in plant regeneration and genetic transformation. Mol. Plant Microbe Interact. 26, 1359–1365 (2013).
Google Scholar
Hallmark, H. T. & Rashotte, A. M. Review—cytokinin response factors: responding to more than cytokinin. Plant Sci. 289, 110251 (2019).
Google Scholar
Rashotte, A. M., Carson, S. D. B., To, J. P. C. & Kieber, J. J. Expression profiling of cytokinin action in Arabidopsis. Plant Physiol. 132, 1998–2011 (2003).
Google Scholar
Šimášková, M. et al. Cytokinin response factors regulate PIN-FORMED auxin transporters. Nat. Commun. 6, 8717 (2015).
Google Scholar
Zwack, P. J., Compton, M. A., Adams, C. I. & Rashotte, A. M. Cytokinin response factor 4 (CRF4) is induced by cold and involved in freezing tolerance. Plant Cell Rep. 35, 573–584 (2016).
Google Scholar
Yang, L. et al. Genetic mapping and regional association analysis revealed a CYTOKININ RESPONSE FACTOR 10 gene controlling flowering time in Brassica napus L. Ind. Crops Prod. 193, 116239 (2023).
Google Scholar
Mason, M. G. et al. Multiple type-B response regulators mediate cytokinin signal transduction in Arabidopsis. Plant Cell 17, 3007–3018 (2005).
Google Scholar
Kiba, T., Aoki, K., Sakakibara, H. & Mizuno, T. Arabidopsis response regulator, ARR22, ectopic expression of which results in phenotypes similar to the wol cytokinin-receptor mutant. Plant Cell Physiol. 45, 1063–1077 (2004).
Google Scholar
Raines, T. et al. The cytokinin response factors modulate root and shoot growth and promote leaf senescence in Arabidopsis. Plant J. 85, 134–147 (2016).
Google Scholar
Fessner, N. D. Enzymatic strategies for next-generation DNA synthesis: boosting efficiency and overcoming secondary structures. ACS Catal. 15, 16106–16114 (2025).
Google Scholar
Freschlin, C. R., Yang, K. K. & Romero, P. A. Scalable and cost-efficient custom gene library assembly from oligopools. Preprint at bioRxiv https://doi.org/10.1101/2025.03.22.644747 (2025).
Barth, S., Goerlich, R. & Schnabl, H. Selection and enrichment of differentially labeled plant protoplasts. J. Biochem. Biophys. Methods 29, 83–86 (1994).
Google Scholar
Dörr, I., Miltenyi, S., Salamini, F. & Uhrig, H. Selecting somatic hybrid plants using magnetic protoplast sorting. Bio/Technology 12, 511–515 (1994).
Google Scholar
Veerabagu, M. et al. The Arabidopsis B-type response regulator 18 homomerizes and positively regulates cytokinin responses. Plant J. 72, 721–731 (2012).
Google Scholar
To, J. P. C. et al. Cytokinin regulates type-A Arabidopsis response regulator activity and protein stability via two-component phosphorelay. Plant Cell 19, 3901–3914 (2007).
Google Scholar
Ren, B. et al. Genome-wide comparative analysis of type-A Arabidopsis response regulator genes by overexpression studies reveals their diverse roles and regulatory mechanisms in cytokinin signaling. Cell Res. 19, 1178–1190 (2009).
Google Scholar
Li, X. et al. CRF12 specifically regulates the flowering time of Arabidopsis thaliana under non-inductive conditions. Plant J. 121, e17257 (2025).
Google Scholar
Cody, W. B., Scholthof, H. B. & Mirkov, T. E. Multiplexed gene editing and protein overexpression using a tobacco mosaic virus viral vector. Plant Physiol. 175, 23–35 (2017).
Google Scholar
Weiss, T. et al. Viral delivery of an RNA-guided genome editor for transgene-free germline editing in Arabidopsis. Nat. Plants 11, 967–976 (2025).
Google Scholar
Beltrán, J. et al. Rapid biosensor development using plant hormone receptors as reprogrammable scaffolds. Nat. Biotechnol. 40, 1855–1861 (2022).
Google Scholar
Tang, B. et al. SALICYLIC ACID SENSOR1 reveals the propagation of an SA hormone surge during plant pathogen advance. Science 390, 188–194 (2025).
Google Scholar
Kaseniit, K. E. et al. Modular, programmable RNA sensing using ADAR editing in living cells. Nat. Biotechnol. 41, 482–487 (2023).
Google Scholar
Huppertz, M.-C. et al. Recording physiological history of cells with chemical labeling. Science 383, 890–897 (2024).
Google Scholar
Brophy, J. A. N. et al. Synthetic genetic circuits as a means of reprogramming plant roots. Science 377, 747–751 (2022).
Google Scholar
Gao, X. J., Chong, L. S., Kim, M. S. & Elowitz, M. B. Programmable protein circuits in living cells. Science 361, 1252–1258 (2018).
Google Scholar
Steinberger, A. R. & Voytas, D. F. Virus-induced gene editing free from tissue culture. Nat. Plants 11, 1241–1251 (2025).
Google Scholar
Qiao, J.-H. et al. Transgene- AND tissue culture-free heritable genome editing using RNA virus-based delivery in wheat. Nat. Plants 11, 1252–1259 (2025).
Google Scholar
Rogers-Broadway, K.-R. & Karteris, E. Amplification efficiency AND thermal stability of qPCR instrumentation: current landscape AND future perspectives. Exp. Ther. Med. 10, 1261–1264 (2015).
Google Scholar
Liu, D. et al. Validation of reference genes for gene expression studies in virus-infected Nicotiana benthamiana using quantitative real-time PCR. PLoS ONE 7, e46451 (2012).
Google Scholar
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 25, 402–408 (2001).
Google Scholar