Digital twins of ex vivo human lungs enable accurate and personalized evaluation of therapeutic efficacy – Nature Biotechnology
Human lung characteristics for DT development
The clinical profiles of the human lungs used to develop the DT models are summarized in Supplementary Table 1. A total of 951 isolated lungs assessed for transplant suitability using the ex vivo circuit were included in this study (Supplementary Fig. 2). Multimodal lung function was evaluated on the EVLP circuit for at least 3 h. The study population represented lungs from disease-free organ donors (that is, no lung-specific diseases or chronic illnesses), with a mean age of 46 ± 16 years and 64% of the cohort was male. The average lung volume was 6.5 ± 1.3 L, which is representative of a typical adult population26. Donor cause of death was primarily due to anoxia (40%) or cerebrovascular injury (32%). Supplementary Table 2 provides a comprehensive list of all lung assessments, including sampling modalities, measurement frequencies and quantification methods, incorporated into the ex vivo human lung DT.
DTs of human lungs accurately forecast human lung function
Lung physiology
DTs of ex vivo human lungs, composed of hybrid physics-informed and data-driven models, accurately forecasted thirteen physiological parameters (Table 1). It is important to note that the static and dynamic DT approaches are different from the metrics of static and dynamic compliance used in this study. Lung compliance is an important assessment of lung physiology as a measure of the ability of the lung to stretch in response to the pressure changes during breathing. There are two measures of lung compliance: static compliance, obtained when airflow is paused, and dynamic compliance, estimated during ventilation. The lung’s capacity to stretch and expand was assessed using physics-based computations of lung mechanics, including dynamic compliance, mean and peak airway pressure and expiratory tidal volume. These measurements were derived from established mechanical equations applied to each breath of the ex vivo lung using high-resolution (100 Hz) ventilator flow and pressure data (Supplementary Fig. 3) and were accurately predicted through integration into a data-driven gated recurrent unit (GRU) model. The mean absolute error (MAE) and mean absolute percentage error (MAPE) values reported for each of the per-breath parameters in Table 1 represent the average MAE and MAPE across 50 forecasted breaths for all cases included in the analysis. We observed that the DT-predicted values deviated from the empirical (ground truth) values by an average of 2–11% (Table 1). Other physiological measurements, such as gas-exchange ability (for example, partial pressures of O2 and CO2), were observed hourly and forecasted using a data-driven XGBoost model, achieving MAPEs of 4–13% (Table 1). Notably, the DT predicted lung edema, a key factor in the safety profile of many therapeutics, within approximately 30 ml of observed values (Table 1).
For all physiological parameters, the dynamic DT demonstrated similar or improved predictive accuracy compared to the static digital lung approach (Table 1). However, the static digital lung provided multiple pathways to predict future lung function: directly from baseline or using predicted values at recently available time points that were used to constantly recalibrate the model. Extended Data Table 1 shows that a static DT model can also directly forecast second-hour and third-hour assessments from the baseline with high accuracy.
In addition, high-resolution physiological data enabled deep learning-based forecasting across multiple look-back windows (for example, 50, 100, 120, 150 and 170 breaths) for both static and dynamic DT setups (Extended Data Table 2). Notably, the data-driven GRU models showed substantial improvements over a non-ML baseline (for example, linear models) for both static and dynamic DTs, indicating that the GRU models successfully captured multivariate biological patterns from earlier per-breath measurements to predict breath parameters (Extended Data Table 2).
Lung biochemistry
Lung biochemistry was assessed in the perfusate solution during EVLP for several electrolytes (for example, [sodium] and [potassium]) and acid–base indicators (for example, pH and [bicarbonate]). The static and dynamic DTs accurately forecasted all seven biochemical evaluations with an MAPE of 0.83–13% and 0.46–7.2%, respectively (Table 1). Similar to physiological assessments, the dynamic digital lung approach demonstrated improved forecasting performance across all parameters. Notably, the dynamic DT dramatically improved the acid–base prediction MAPEs (pH, from 0.85% to 0.46%; base excess, from 5.5% to 2.7%; bicarbonate, from 13% to 7.2%) compared to the static digital lung (Table 1).
Lung omics
Metabolomic biomarkers
Cellular metabolism was modeled by the DT using glucose and lactate levels in EVLP perfusate. Overall, both the static and the dynamic approaches demonstrated good performance in forecasting glucose (within 0.3–0.5 mM) and lactate (within 0.5–0.9 mM) levels (Table 2). As previously observed, additional data measurements leveraged by the dynamic digital lung models improved glucose and lactate simulation accuracy, resulting in MAPE values of 6.9% for lactate and 4.9% for glucose (Table 2).
Transcriptomics
The ability to forecast transcriptomic changes in a lung can be enabled by tissue biopsies collected before and after EVLP. Thus, using the pre-EVLP biopsy as a transcriptomic baseline, a data-driven approach leveraging ex vivo lung function measurements (Fig. 1c) enabled the DT to predict the post-EVLP transcriptome. Table 2 highlights that ex vivo human lung DTs can accurately forecast transcriptomic changes in lung disease-related pathways27,28,29,30,31,32,33,34,35 with high accuracy (MAPE: 1–3%). For example, both the static and the dynamic DT models accurately forecasted the gene enrichment score of the TGFβ signaling pathway, hypoxia pathway and p53 pathway (MAPE: 0.68–0.97%). Additional gene sets were observed to have similar forecasting results (Extended Data Table 3). Unlike the lung physiology and biochemistry parameters, for the prediction of transcriptomic changes, the dynamic DT did not always perform better than the static digital lung (Table 2).
Enrichment scores are unitless measurements that reflect the activity level of the biological processes associated with genes in a particular gene set and MAPE values can be less clinically representative and interpretable. Therefore, a key aspect of transcriptomic analysis, beyond knowing the gene set enrichment score, is identifying temporal changes in gene set enrichment for a given lung. Ex vivo human lung DTs demonstrated an accuracy of 79–100% to correctly identify the temporal changes in gene enrichment (for example, upregulation) (Extended Data Table 4). Notably, the per-case prediction of the hypoxia pathway reached 100% accuracy, suggesting that the DT can accurately predict the directional change of the hypoxia pathway for human lungs on EVLP (Extended Data Table 4).
Proteomic biomarkers
During EVLP, circulating proteins can be analyzed in the perfusate solution. Using an ELISA-based approach, we observed that the dynamic digital lung approach can predict concentrations of inflammatory protein biomarkers with an average error of 23 pg ml−1 for IL-10, 2.5 pg ml−1 for IL-1β, 6.2 ng ml−1 for IL-6 and 0.53 ng ml−1 for IL-8 (Table 2). In the study cohort, we observed larger than expected MAPE values because of the extreme range of protein concentrations with some approaching zero and causing the average percentage error values to be skewed and less representative of the forecasting performance. Notably, the DT showed median MAPE values of 23% for IL-10, 26% for IL-1β, 22% for IL-6 and 37% for IL-8, which were more closely aligned with the forecasting accuracy of other parameters.
Lung imaging
The EVLP platform allows imaging of the lungs in isolation, without the confounding factors present in a traditional chest X-ray, such as the heart, chest wall and ribs. X-ray images taken after 1 and 3 h of perfusion were acquired and studied for DT development (Supplementary Fig. 4). A convolutional neural network (CNN) approach was used to derive the top ten principal components (PCs) as previously described13,14. A data-driven XGBoost model was able to accurately simulate the third-hour image PCs (Supplementary Table 3). As PC values are unitless, MAE and MAPE are less clinically intuitive for interpreting model results. It has been previously reported that the image PCs are strongly correlated to radiographic findings and diagnoses14. Both static and dynamic digital lung approaches of image PC values revealed strong correlations with important clinical radiology findings (for example, consolidation and infiltration) and diagnoses (for example, aspiration and pneumonia), underscoring the accuracy of the DT and its ability to identify clinically important image features (Table 3).
DTs of human lungs demonstrate accurate predictions with strong benchmark performance
Next, we assessed the accuracy of the DT predictions. One-sample t-tests comparing the mean prediction error against zero (that is, representing perfect prediction) found no statistically significant deviation across key lung functional parameters or model configurations (Supplementary Table 4). These findings indicate the absence of systematic bias, as the digital lung models did not consistently overestimate or underestimate their forecasted values. We then evaluated the DT relative to an important design requirement to outperform a traditional control group by comparing its performance to real-world variability using an empirical cohort. Random sampling of historical EVLP cases was used to assess data variability within a traditional control group, establishing a performance benchmark that ex vivo human lung DTs should meet or exceed to demonstrate clinical utility. The absolute error of the DT was consistently lower than the coefficient of variation (%CV) observed in an empirical control group of five lungs, reflecting typical24,25 EVLP study designs (Extended Data Table 5). This narrower error distribution of the DT demonstrates greater precision and robustness than traditional small-cohort control groups.
DT model validation
We then sought to validate the accuracy and calibration of the ex vivo lung DTs by assessing performance across two distinct cohorts.
First, we assessed the DT on a cohort of n = 50 simulated lung profiles on the basis of realistic, physiologically relevant demographics features using a k-nearest neighbor (KNN) approach (Methods)36. The MAEs and MAPEs observed in the simulated cohort were consistent with the performance metrics observed during model training (Supplementary Tables 5–7). The simulated lung cohort generated using a KNN approach based on a historical cohort serves as an illustration for the static and dynamic DT model that could represent a comprehensive, multimodal DT. Figure 2 provides an illustrative example to demonstrate the complete baseline and forecasting profile of lung physiology, biochemistry, omics and imaging for a human lung on the ex vivo platform.
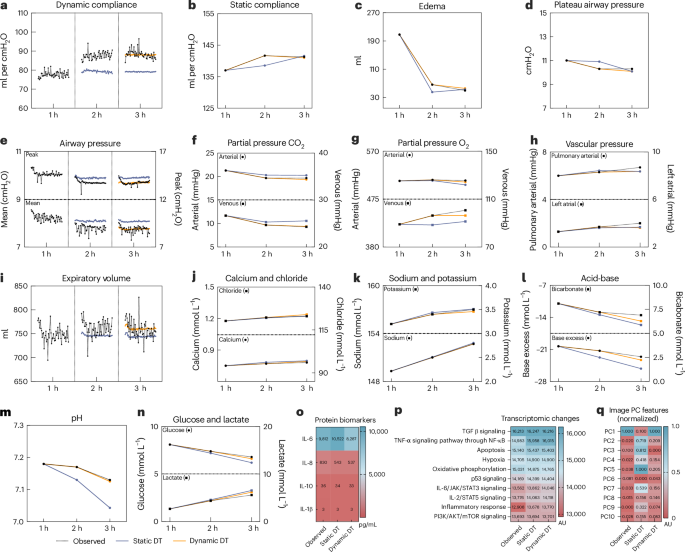
a–q, Static (blue) and dynamic (orange) multimodal forecasting DTs of a simulated human lung on EVLP, using baseline functional data (dashed black lines) for lung physiology (a, dynamic compliance; b, static compliance; c, edema; d, plateau airway pressure; e, peak and mean airway pressures; partial pressure of CO2 (f) and O2 (g); h, pulmonary arterial and leftatrial pressure; i, expiratory volume), lung biochemistry (j, calcium and chloride; k, sodium and potassium; l, bicarbonate and base excess; m, pH), lung omics (n, metabolomic biomarkers (glucose and lactate); o, protein biomarkers (IL-6, IL-8, IL-10, IL-1β)), transcriptomics (p, gene enrichment scores for lung disease-related pathways) and lung imaging (q, image PC values (normalized) derived from the third-hour X-ray image).
Source data
We then evaluated the DT on an independent test dataset of 45 clinical ex vivo human lungs, distinct from the training cohort, to assess the DT robustness and generalizability to unseen data. Across the physiological, biochemical and image-derived parameters, MAE and MAPE values of the test dataset were consistent with DT performance observed in out-of-fold predictions from k-fold cross-validation on training data (Extended Data Tables 6 and 7). The observed agreement between training and testing performance demonstrates effective DT calibration during training and sustained predictive accuracy when applied to clinical cases.
To enhance clinical utility and facilitate seamless adoption of human lung DTs, the models developed in this study were integrated and deployed using a web-based application (https://dt-lung.streamlit.app/).
DTs of human lungs enable the evaluation of therapeutic efficacy and safety
To expand on the findings of more efficient clinical studies using DTs, real-world evidence was evaluated for human lungs treated with the therapeutic alteplase37. Alteplase, a tissue-type plasminogen activator that is often used clinically to lyse clots, has thrombolytic effects and reduces vascular resistance38,39. During the multiorgan recovery process, lungs are particularly vulnerable to developing pulmonary embolism (PE)—blood clots in the pulmonary vasculature, which can severely impair lung function if left untreated40. While not all lungs develop PE, EVLP provides an opportunity to assess the risk of PE and administer targeted treatment using thrombolytics, such as alteplase37.
In cases of clinical PE, vascular blockages typically result in elevated pulmonary vascular pressure. Treatment with alteplase dissolves these blockages, thereby reducing vascular pressure. Consequently, lungs that respond to alteplase are expected to demonstrate improved function (that is, lower vascular pressure), potentially leading to favorable clinical outcomes, such as shorter recovery times for transplant recipients. Conversely, lungs that do not respond effectively to alteplase are likely to experience worse clinical outcomes. Historically, evaluating therapeutic efficacy in an individual lung has been challenging because of the absence of a paired control for comparison.
On the basis of our clinical EVLP protocol, alteplase was administered after baseline data (that is, first-hour data) was acquired. Using first-hour baseline lung data as input, the DT simulated counterfactual untreated outcomes for alteplase (that is, the outcome that would have occurred had the lung not received therapy), enabling causal inference to quantify individual treatment effects from forecasted versus observed data. Thus, the therapeutic efficacy and safety profiles of alteplase-treated lungs were evaluated using a DT approach (that is, each treated lung was compared to its corresponding untreated DT). Pulmonary arterial pressure (PAP) was evaluated as the primary efficacy endpoint for therapeutic benefit, while an estimate of edema was used as a metric to assess the drug’s safety profile. Figure 3a,b illustrates gross macroscopic evidence of PE in a human lung. Figure 3c,d demonstrates therapeutic efficacy (lowered PAP) and safety (no additional edema formation) of alteplase for a treated lung versus its corresponding DT.

a,b, Representative images of human lungs with suspicion with PE (source: UHN) (a, image of human lungs with evidence of possible vascular obstructions; b, image of a blood clot obtained during retrograde flushing of the lungs in a). c,d, Representative data from measurements of the observed (black solid square) and forecasted (red dashed square) PAP and observed (black solid circle) and forecasted (red dashed circle) edema formation for alteplase-treated cases 1 (c) and 4 (d). e–h, Evaluation of the efficacy of alteplase to modify PAP 1 h (e,f) and 2 h (g,h) after administration using a conventional control cohort (e,g) or DT cohort (f,h) (n = 6). i–l, Evaluation of the safety of alteplase to reduce edema formation 1 h (i,j) and 2 h (k,l) after administration using a conventional control cohort (i,k) or DT cohort (j,l) (n = 6). Box plots depict the median, upper and lower quartiles and range. Individual data points (n = 6) shown in e–l denote six independent ex vivo human lungs treated with alteplase. Statistical analysis was conducted using a two-sided Mann–Whitney test (a) or two-sided Wilcoxon matched-pairs signed rank test (b).
Source data
To further evaluate the benefits of the DT in preclinical research, pooled statistical analyses were performed (Fig. 3e–l and Extended Data Table 8). Donor lung suitability for transplantation was determined clinically by surgical assessment during ex vivo evaluation. For lungs with function determined suitable for transplantation at the end of EVLP (n = 6), we observed a similar safety and efficacy response to alteplase (Extended Data Table 8). For lungs that were not suitable for transplantation and associated with poor outcomes (n = 8), the lungs did not appear to respond positively to alteplase (Extended Data Table 8). A conventional cohort-based analysis compared alteplase-treated lungs to a randomly selected control group of EVLP lungs (n = 6). This traditional study design revealed no significant differences in PAP observed at 1 h (P = 0.36) or 2 h (P = 0.78) after treatment or in edema at 1 h (P = 0.21) or 2 h (P = 0.78) after treatment (Fig. 3e,g,i,k). In contrast, the DT approach was leveraged as a personalized control, enabling paired statistical analyses between observed and forecasted PAP and edema values. Most notably, the DT analysis revealed significantly lower PAP (P = 0.031) 2 h after treatment using a two-sided Wilcoxon matched-pairs signed rank test (Fig. 3h and Extended Data Table 8). The effects of alteplase were only observed in lungs that were deemed suitable for transplantation with good postoperative outcomes (Extended Data Table 8). In lungs that were unsuitable for transplant with poor outcomes, there was no observed effect of alteplase (Extended Data Table 8). Compared to their respective DTs, lungs associated with unsuitable clinical outcomes did not show a lowering effect on PAP, indicating an absence of alteplase efficacy (Extended Data Table 8 and Extended Data Fig. 1). In these lungs, PAP recordings after treatment were the same or higher than their DT counterparts, while edema measurements showed no significant changes, confirming safety but no therapeutic benefit on a per-case basis (Extended Data Fig. 2). Traditional cohort-based analyses in lungs associated with poor clinical outcomes confirmed that alteplase treatment did not show signs of improved lung function (Extended Data Table 8 and Extended Data Fig. 1). Significant improvements in lung function after alteplase treatment were observed only in lungs with good clinical outcomes when using the DT approach. These findings would have not been observed using a traditional control cohort. Moreover, while edema values were not significantly different 120 min after treatment in the suitable lung cohort, there was a trend toward the treated lungs showing improved lung function (P = 0.063), contributing to the observed lower edema values 120 min after treatment (Fig. 3l).






